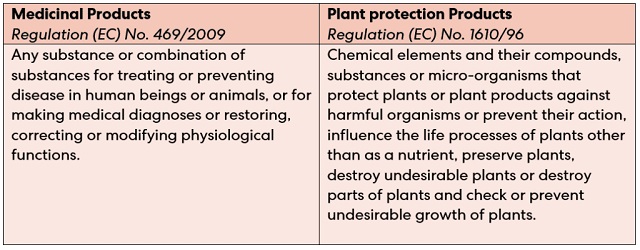
Is article 3 (a) of the Regulation (EC) 469/2009 disappearing into the mist, or is clarity round the corner? - The IPKat
EUROPEAN COMMISSION Brussels, 28.5.2018 COM(2018) 317 final 2018/0161 (COD) Proposal for a REGULATION OF THE EUROPEAN PARLIAMENT
Council Regulation (EEC) No 1768/92 of 18 June 1992 concerning the creation of a supplementary protection certificate for medici
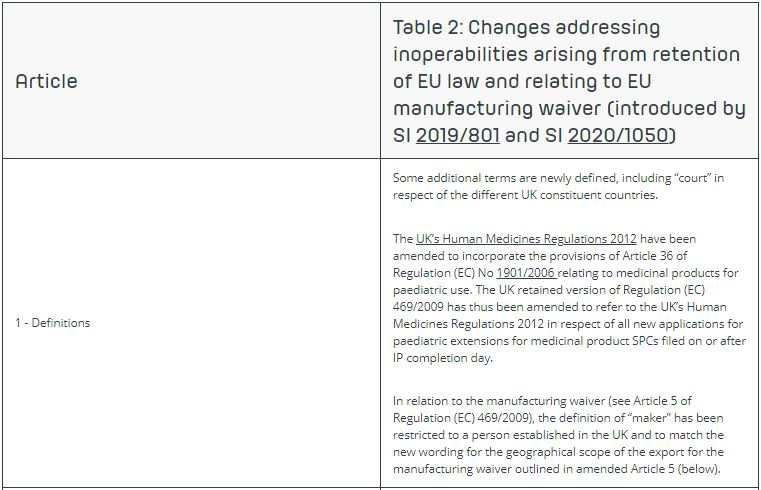
Pursuant to Article 36(1) of Regulation (EC) No 1901/2008 on medicinal products for paediatric use and amending Regulation (EEC)
CJEU refuses UK HC's request for preliminary ruling on SPC applications based on third-party MAs, on account of referred que
GENERAL STATEMENT 2017 – NORWAY – WIPO Assemblies, 57th Series of Meetings, October 2 to 11, 2017 The Norwegian Delegation w
Second medical indication and SPC - Referral to the CJEU for a preliminary ruling on the interpretation of Article 3d) of Regulation (EC) No 469/2009 in the light of the NEURIM judgment (
Field of research: Intellectual Property – Life sciences- Patents- Access to Health Technologies and Innovation. Title: Change