Market Access in Europe: Bridging Regulatory and Health Technology Assessment (HTA) Gaps | Voisin Consulting Life Sciences

Frontiers | Towards a European harmonization of health technology assessment recommendations executive paper of European regulatory conference focused on the EU commission proposal to harmonize HTA

Language derived from the preamble of the EU HTA Regulation regarding... | Download Scientific Diagram
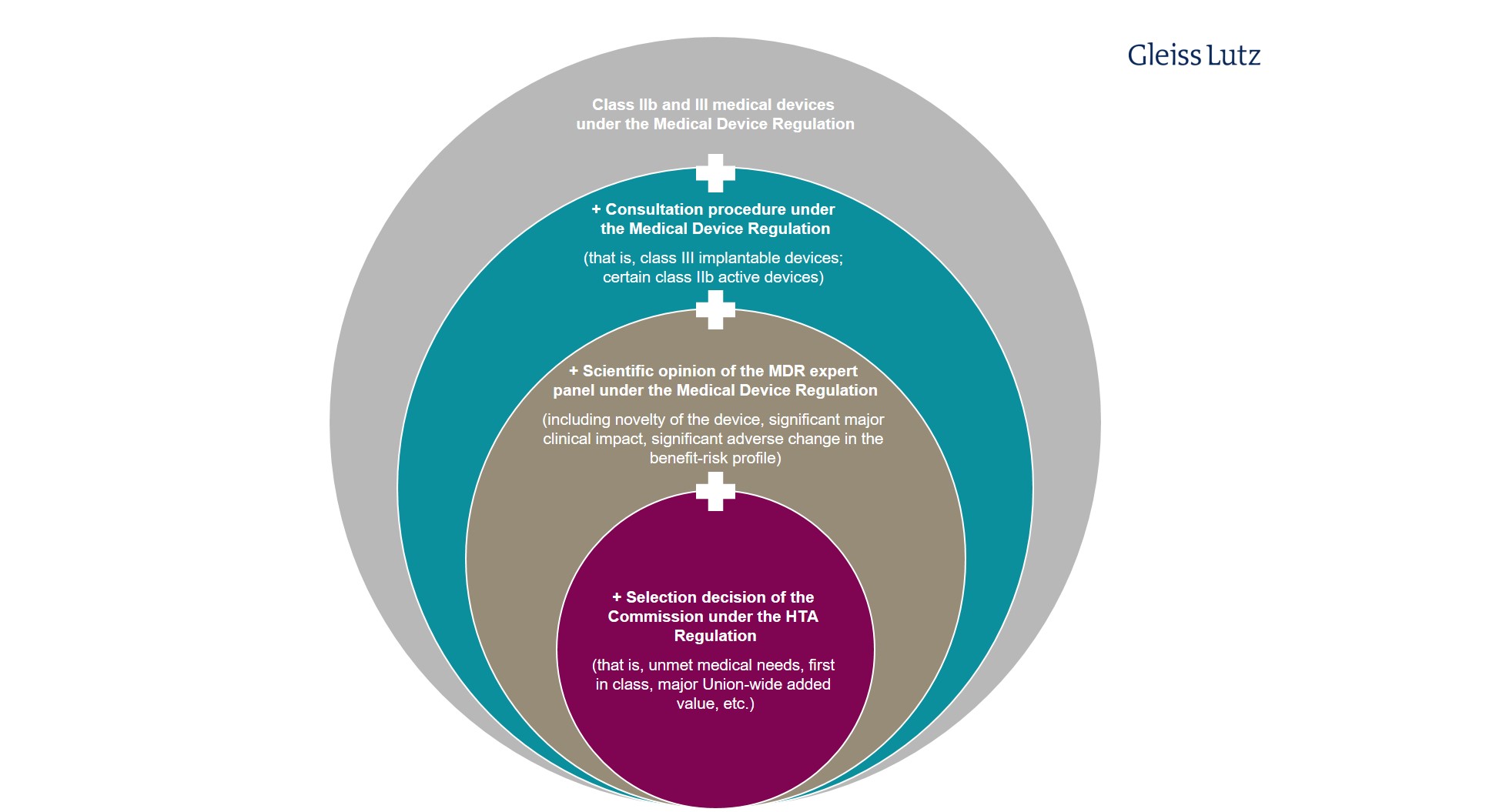
The impact of the EU HTA Regulation: “Booster” for innovative medical devices in Europe? | Gleiss Lutz
The New HTA Regulation: key elements and next steps - Flora Giorgio (B6, DG SANTE, European Commission)

HTA Guide to Quality and Safety Assurance for Tissues and Cells for Patient Treatment | Human Tissue Authority
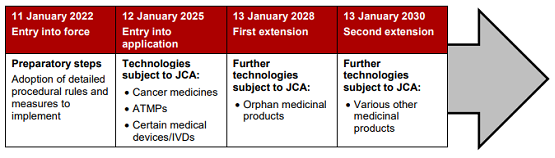
New Regulation on Health Technology Assessment Seeks to Facilitate EU-Level Joint Clinical Assessments From 2025 - Lexology
Aprobado por el PE el borrador de Reglamento de EVALUACION DE TECNOLOGIAS SANITARIAS (HTA) que incluye los productos sanitarios @EU_Health

Medical Writing | Medical decision making and health technology assessment | The regulatory-HTA decision-making interface: What the medical writer should know