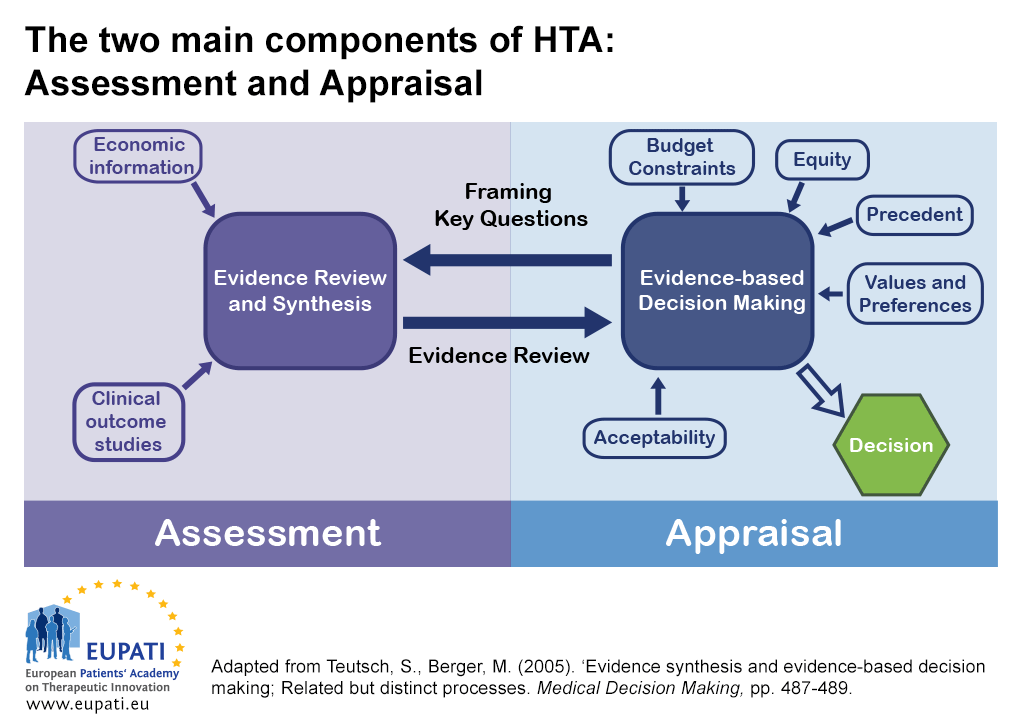
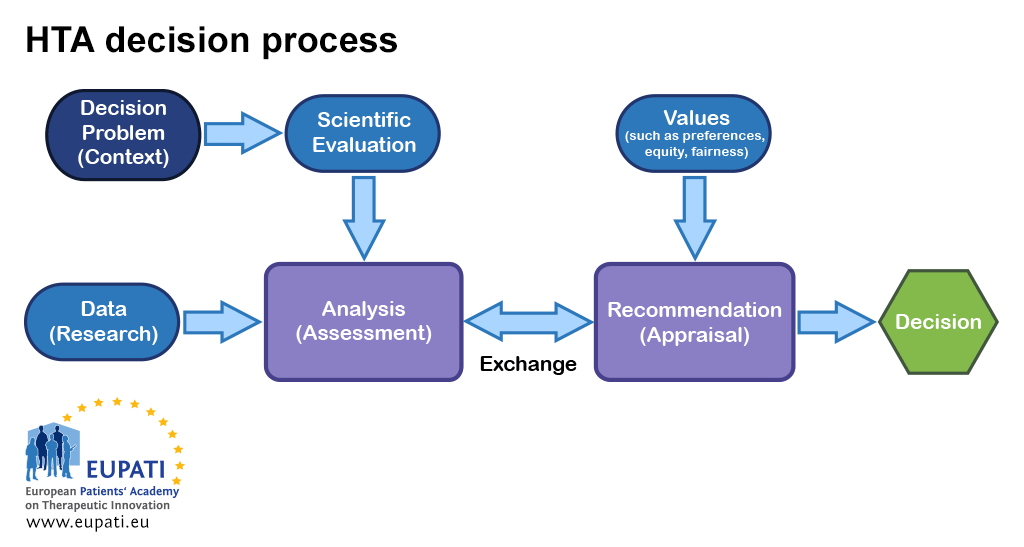
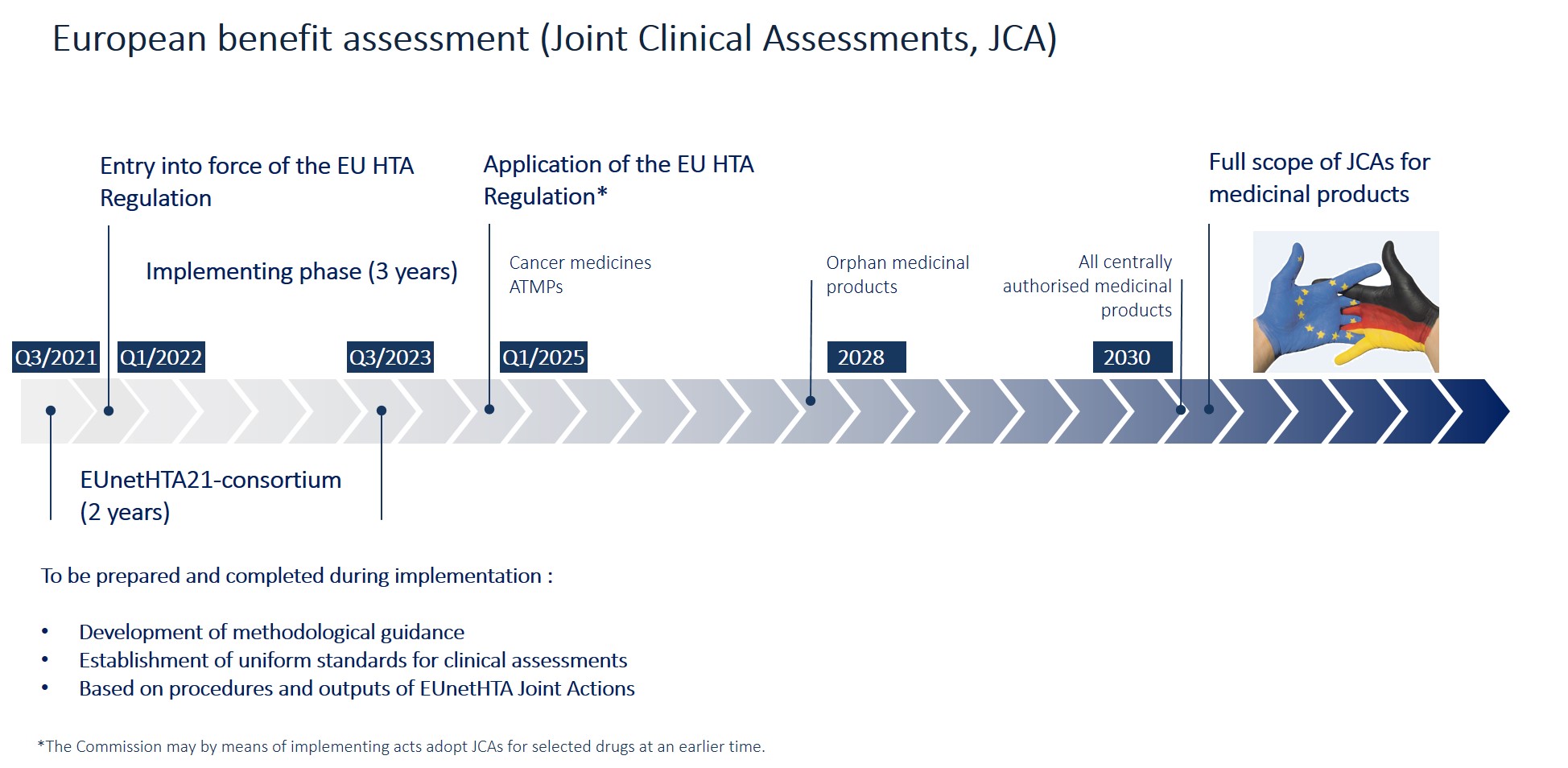
KSR Systematic Reviews al Twitter: "A great overview of #HTA in Europe spanning from #EUR-ASSESS in 1994 to the recent #EU-HTA-Regulation, hopefully leading to the creation of a single #sustainable #EuropeanNetwork #HTA

HTA Evaluación Tecnologia Sanitaria «The Regulation on HTA – What's 22 jun 2022 Bruselas 10h CEST por streaming | Red de Tecnologías Sanitarias y Productos Sanitarios

The impact of the EU HTA Regulation: “Booster” for innovative medical devices in Europe? | Gleiss Lutz

PharmaBoardroom - The New EU Health Technology Assessment Regulation: Building Optimal Regulatory & HTA Collaboration


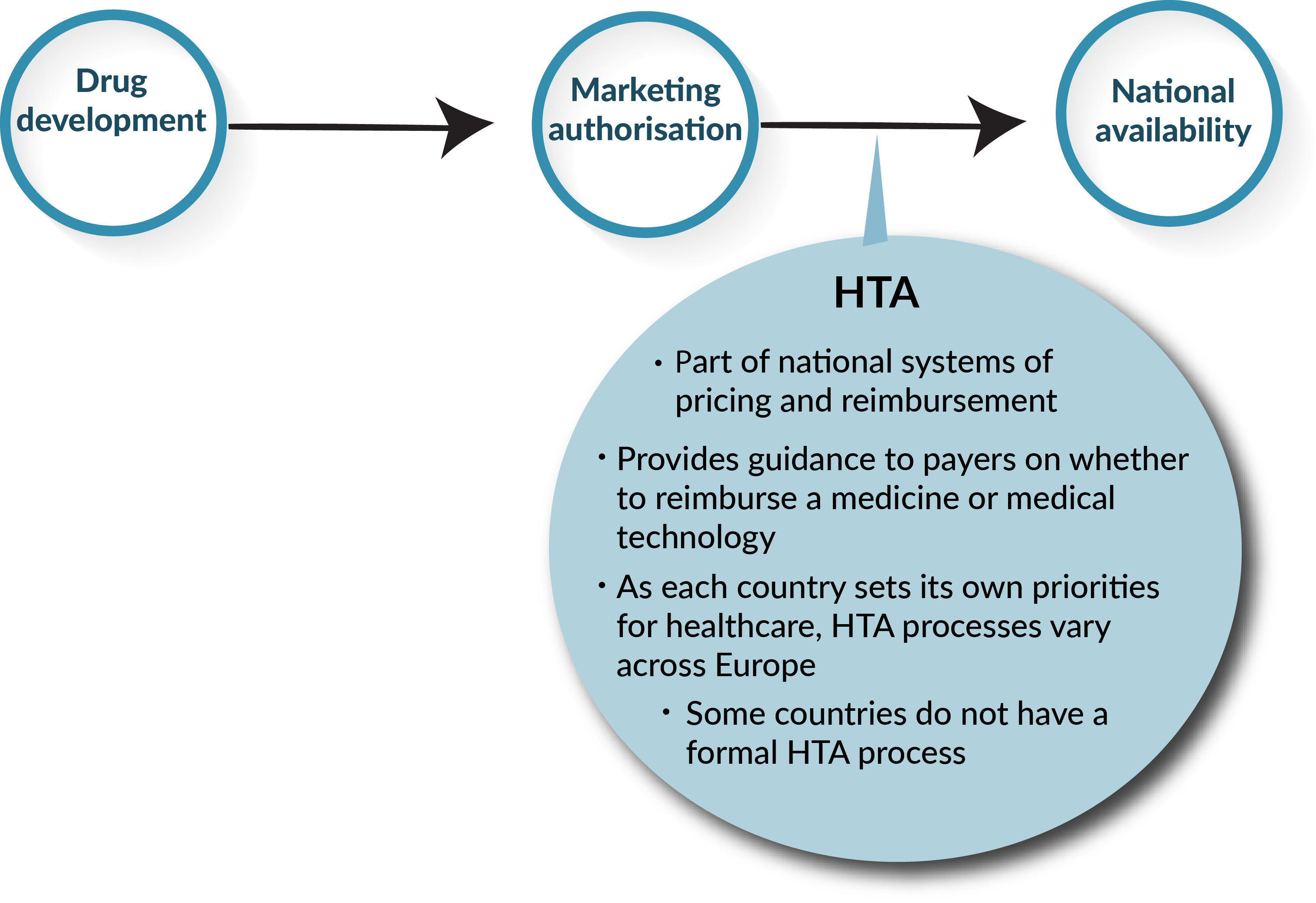





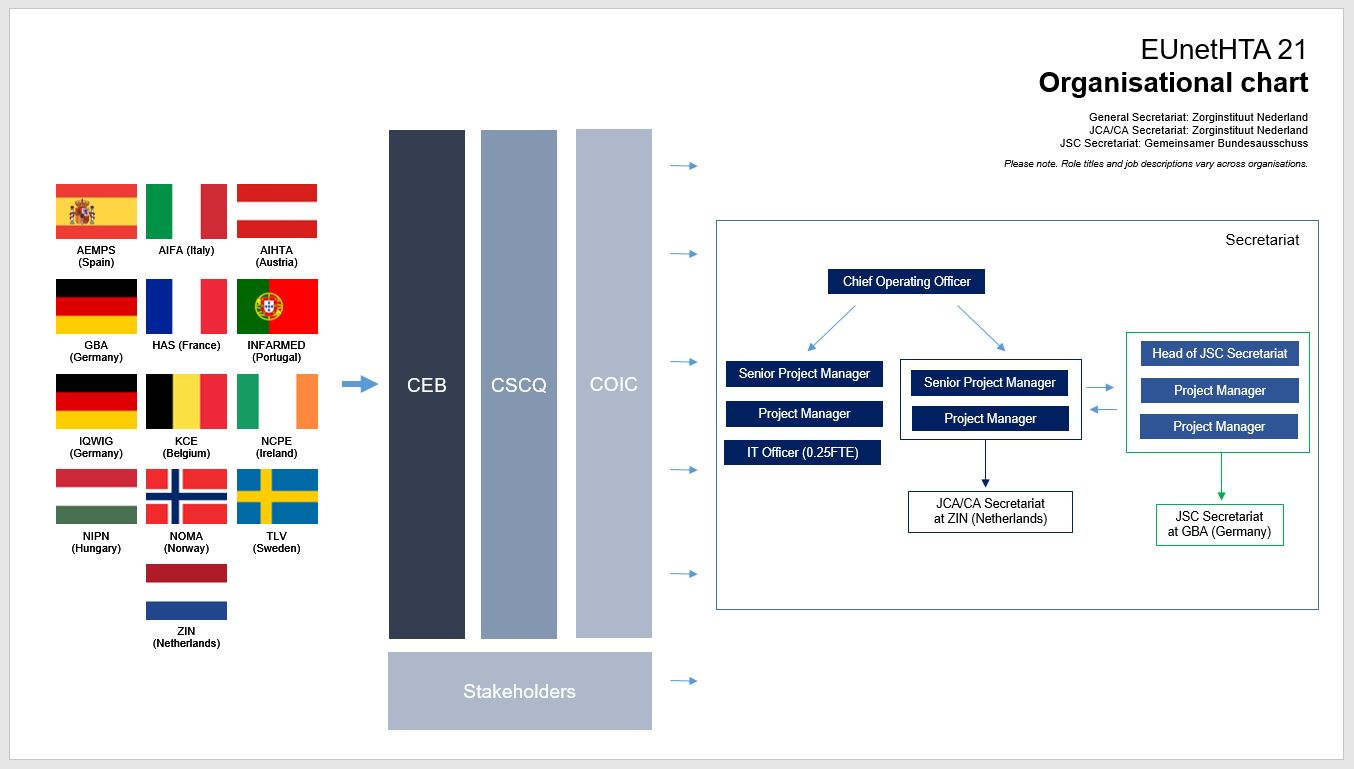
.jpg)