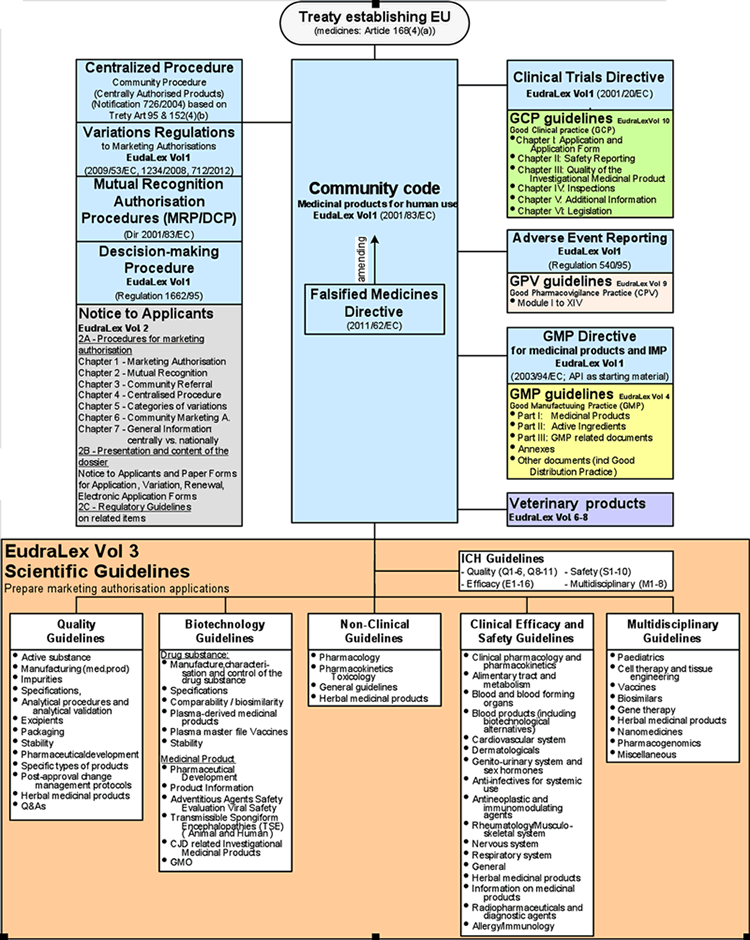
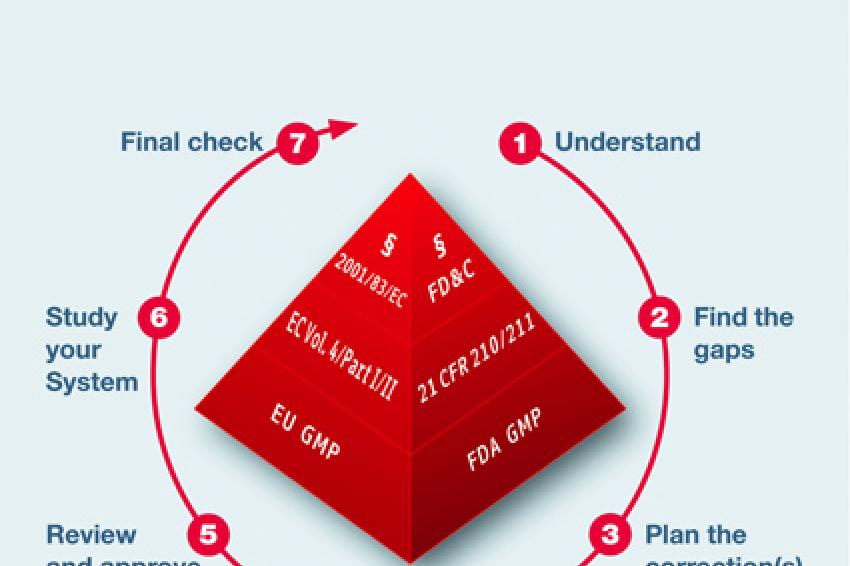
How will the new EU GMP Annex 21 influence the importation of medicinal products? - Blog - Healthmed

A comparison of the salient points in QA and cGMP regulation in the US... | Download Scientific Diagram

Good Manufacturing Practice (GMP) Guidelines: The Rules Governing Medicinal Products in the European Union, EudraLex Volume 4 Concise Reference : Allport-Settle, Mindy J.: Amazon.es: Libros

EudraLex - Volume 4 - Good Manufacturing Practice (GMP) guidelines - Free PDF download | M A N O X B L O G

Book 4C: 2022 Good Manufacturing Practice in the European Union, Refer – Clinical Research Resources, LLC

News about GMP/cGMP - GMP-Verlag: EU: EU Clinical Trials Regulation and new Information System finally in place

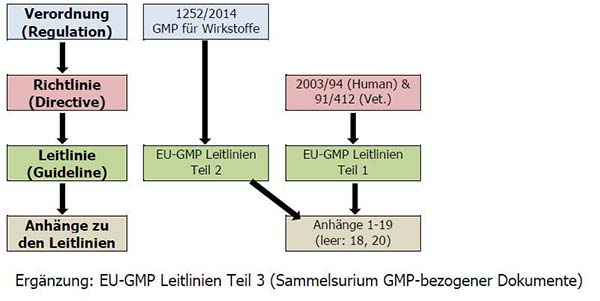
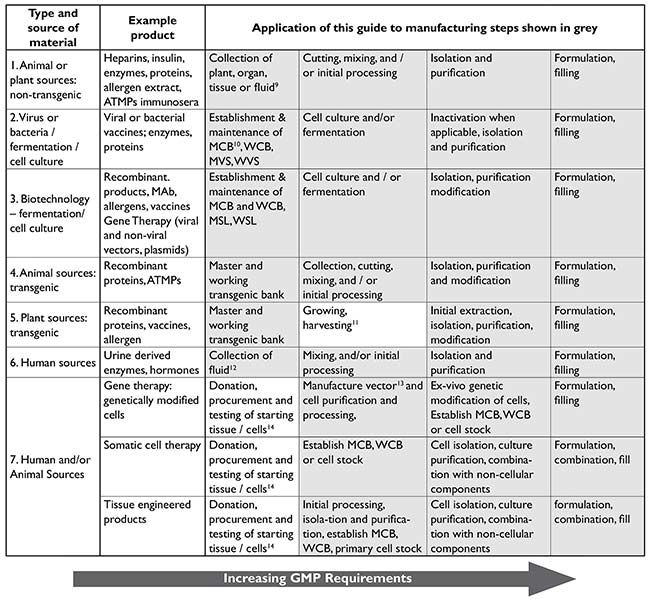
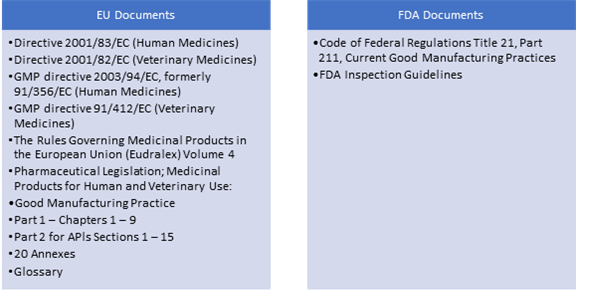
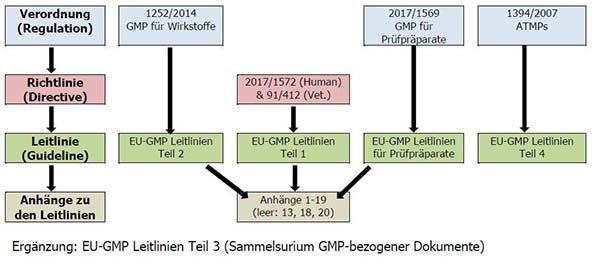
EU GMP and Quality Oversight: How does it work? - GMP- und GXP Experten | Expertsinstitut Beratungs GmbH