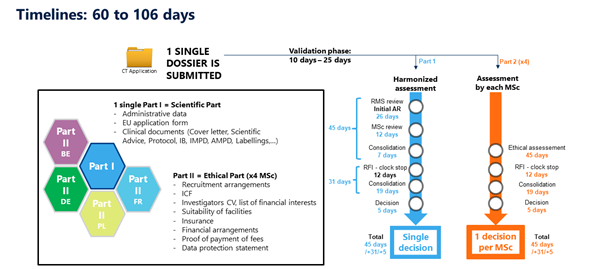
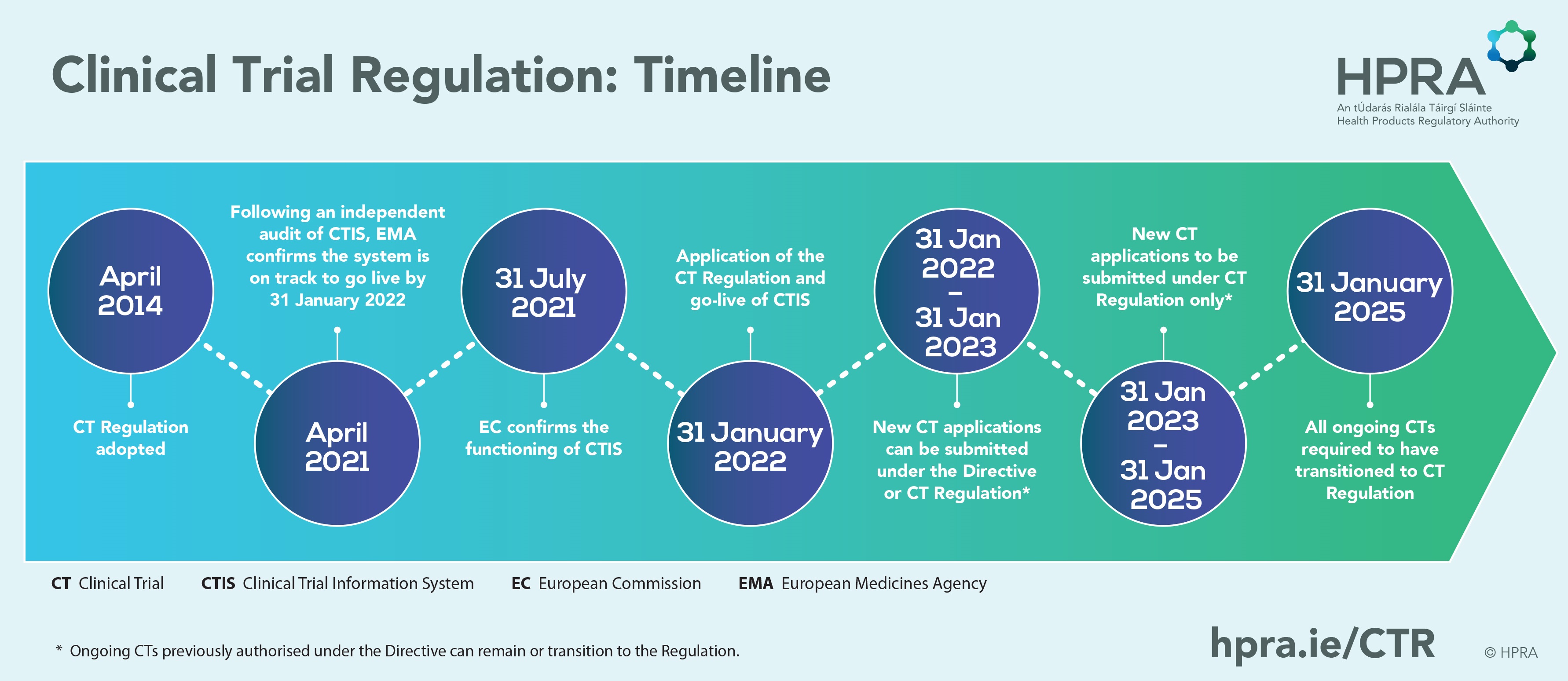
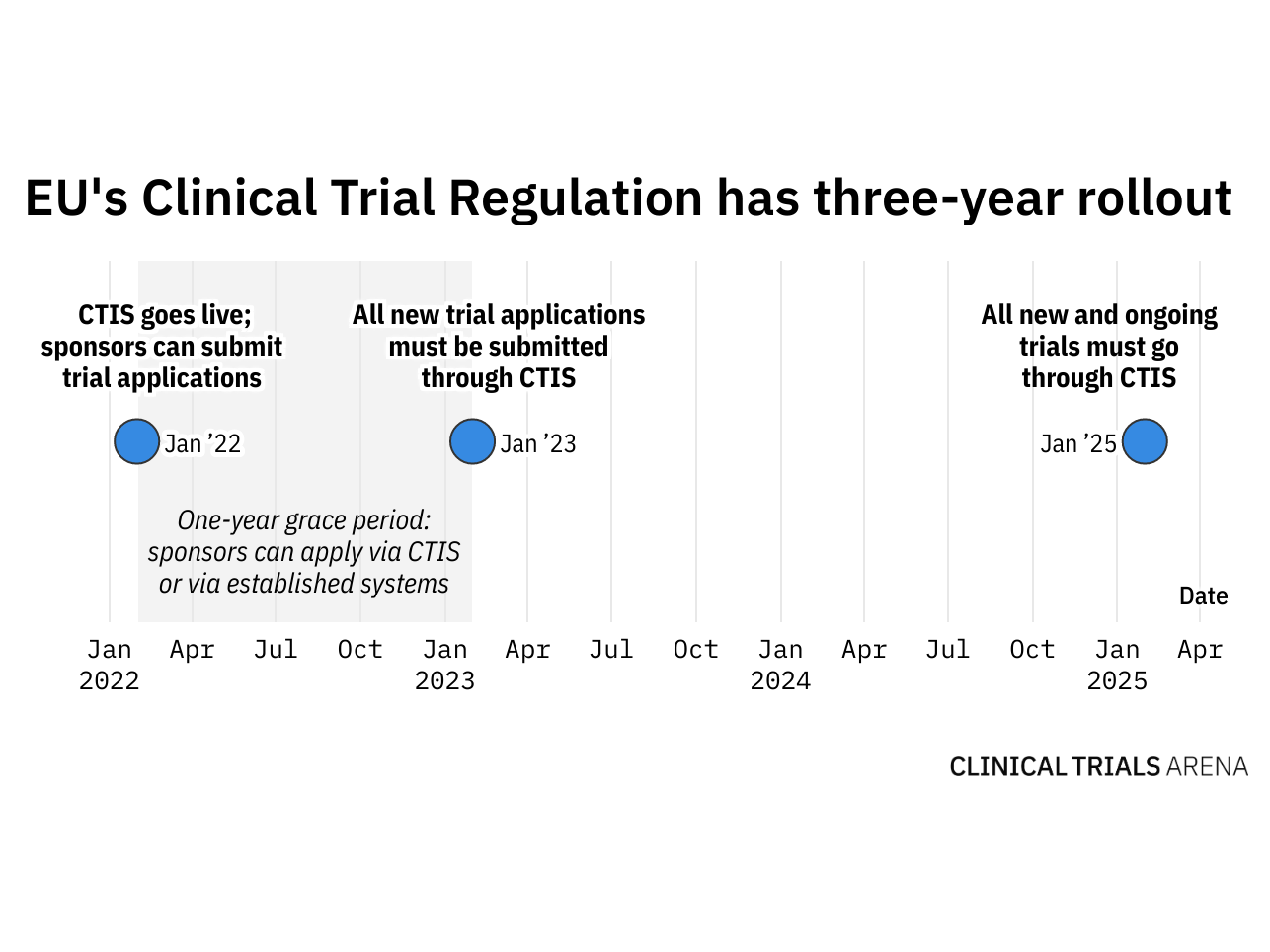
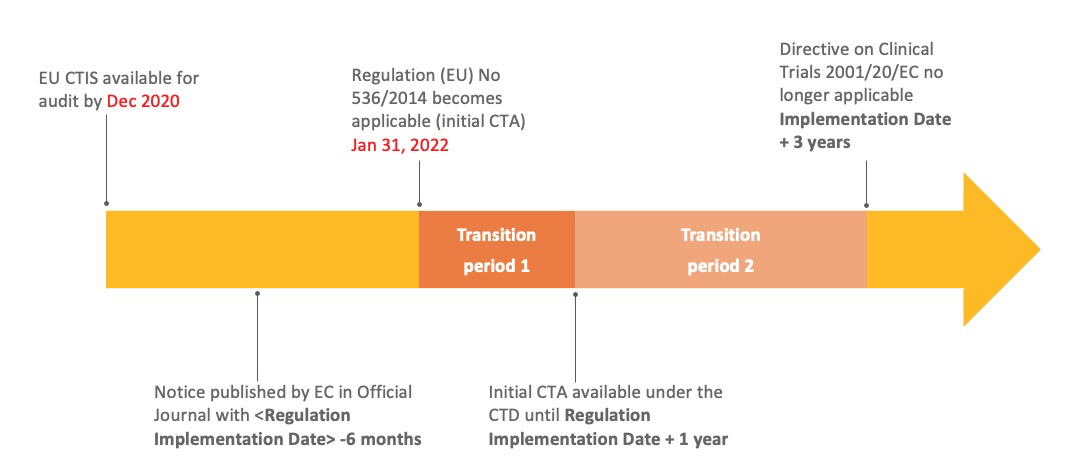
Clinical Trials Information System, as part of the Clinical Trials Regulation to ensure: ✓Higher safety protection for participants ✓Faster access to the most promising safe & effective medical products ✓EU-wide clinical trials

MHRA to streamline clinical trial approvals in biggest overhaul of trial regulation in 20 years - GOV.UK
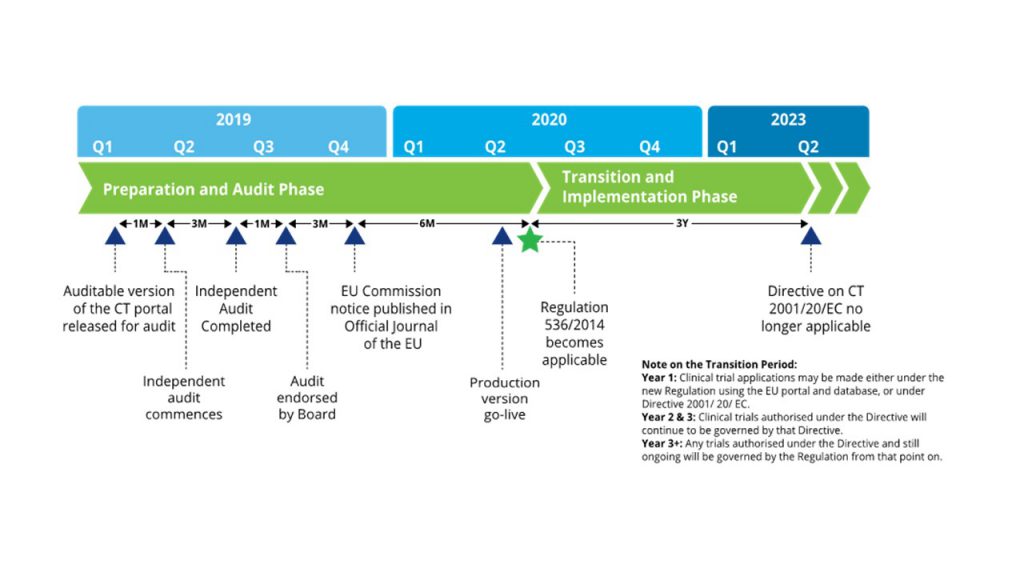
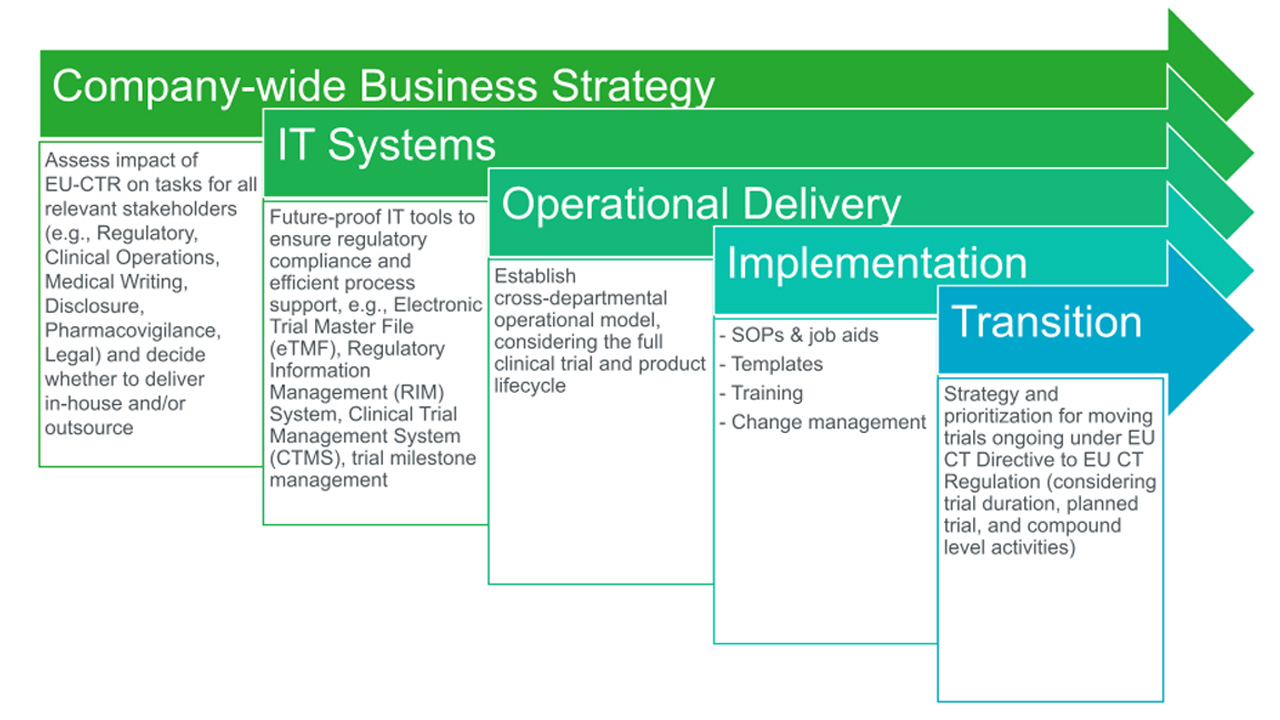
ADVANCED CLINICAL TRIALS THE CLINICAL TRIAL PROCESS: IMPENDING CHANGES IN THE REGULATORY FRAMEWORK - ADVANCED CLINICAL TRIALS










![Withdrawn] Clinical Trials Regulation - GOV.UK Withdrawn] Clinical Trials Regulation - GOV.UK](https://assets.publishing.service.gov.uk/government/uploads/system/uploads/image_data/file/77311/s960_Digital_logo-for-gov-uk_-_Copy.png)