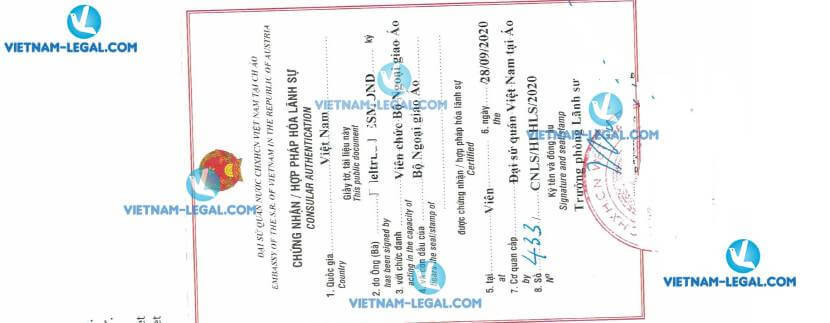
Result of Certificate of a Pharmaceutical Product issued in Austria for use in Vietnam on 28.09.2020 - legalization

Guidance on obtaining electronic certificates of pharmaceutical products and good manufacturing practices (GUI-0024) - Canada.ca

RaMar DXN Products Philippines - GMP CERTIFICATE OF MANUFACTURING FACILITY FROM TGA, AUSTRALIA (Page 2) Accorded to DXN Pharmaceutical Sdn. Bhd. for its high quality,safe, efficacious and availability of products, which fulfill

Pharmaceuticals Quality Commitment and Certifications - Allenge India ( Pharmaceutical Company) (Allenge India Pharma) committed to maintain the highest standards of quality, the products meet relevant pharmacopeial standards and statutory requirements

Indian Regulator extends validity of Certificate of Pharmaceutical Product (COPP) to facilitate Pharmaceutical Industry - API FIRST