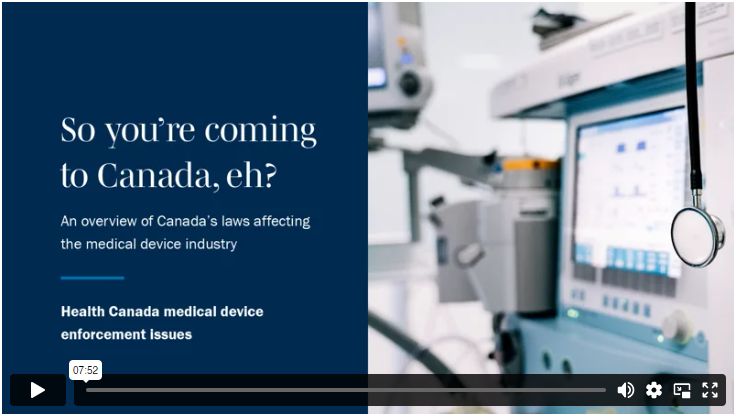
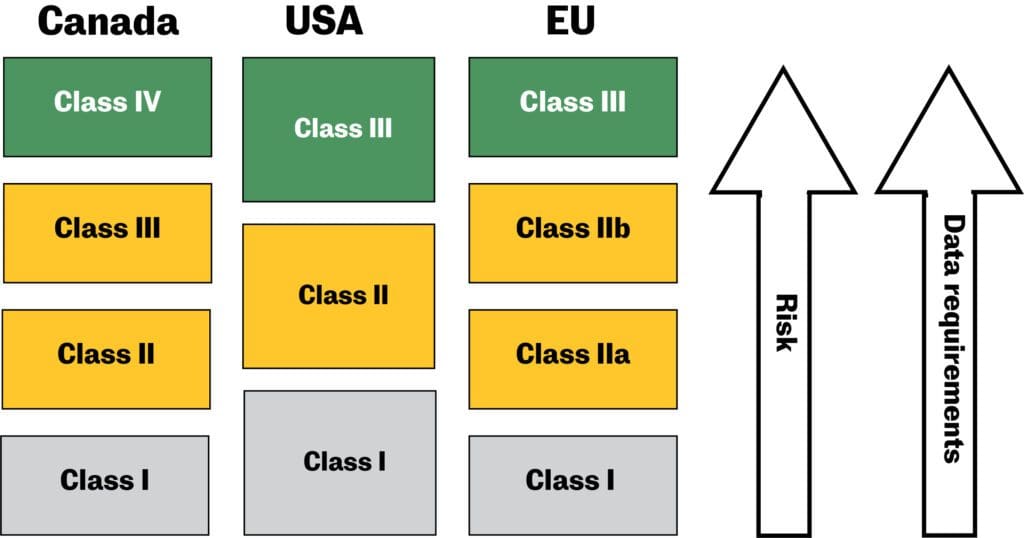
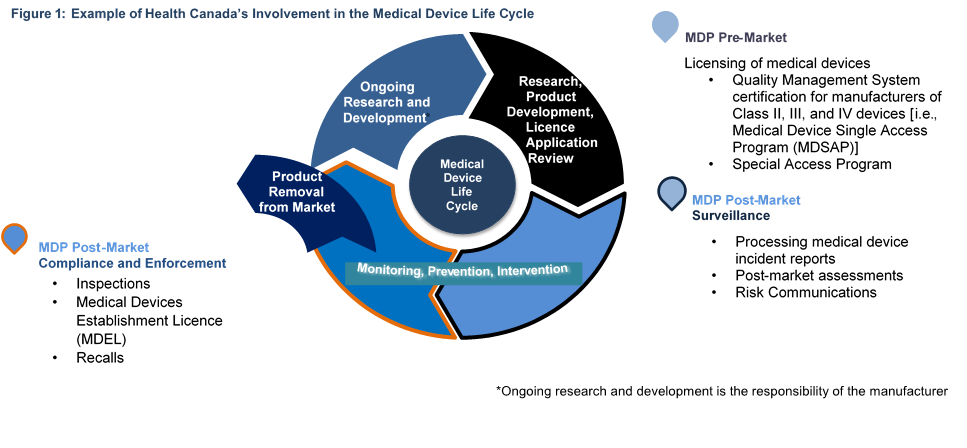
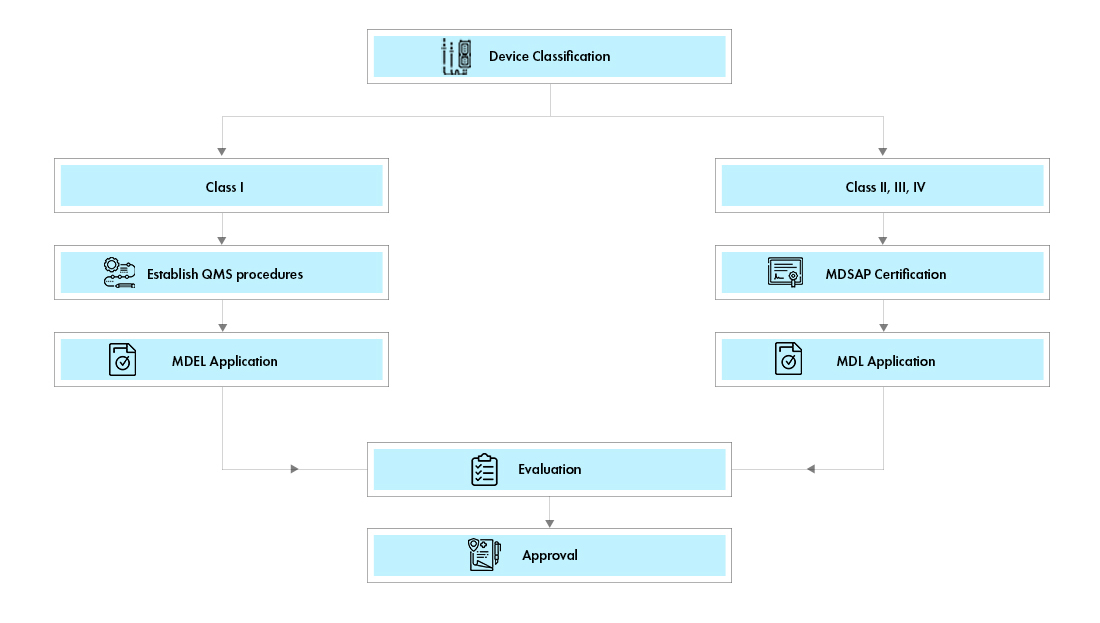
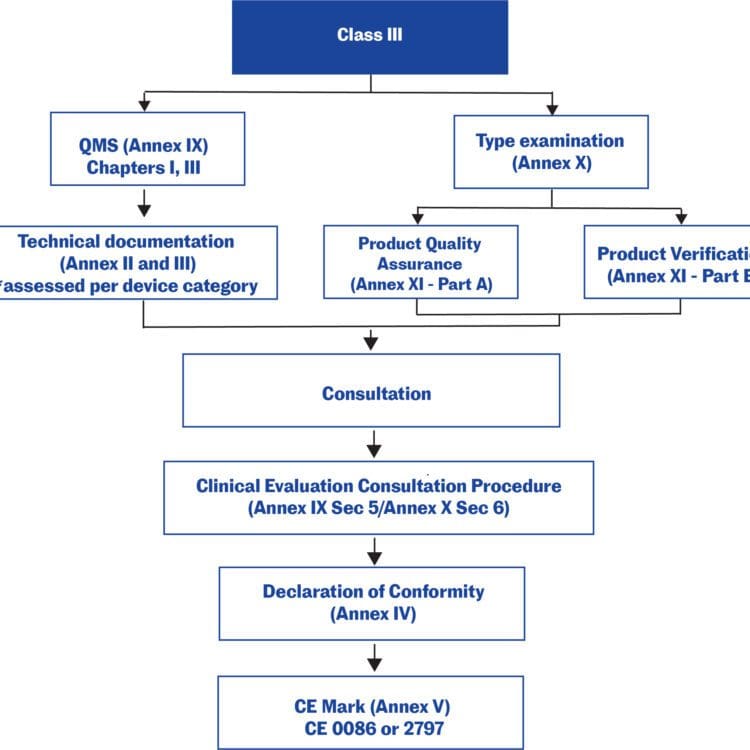
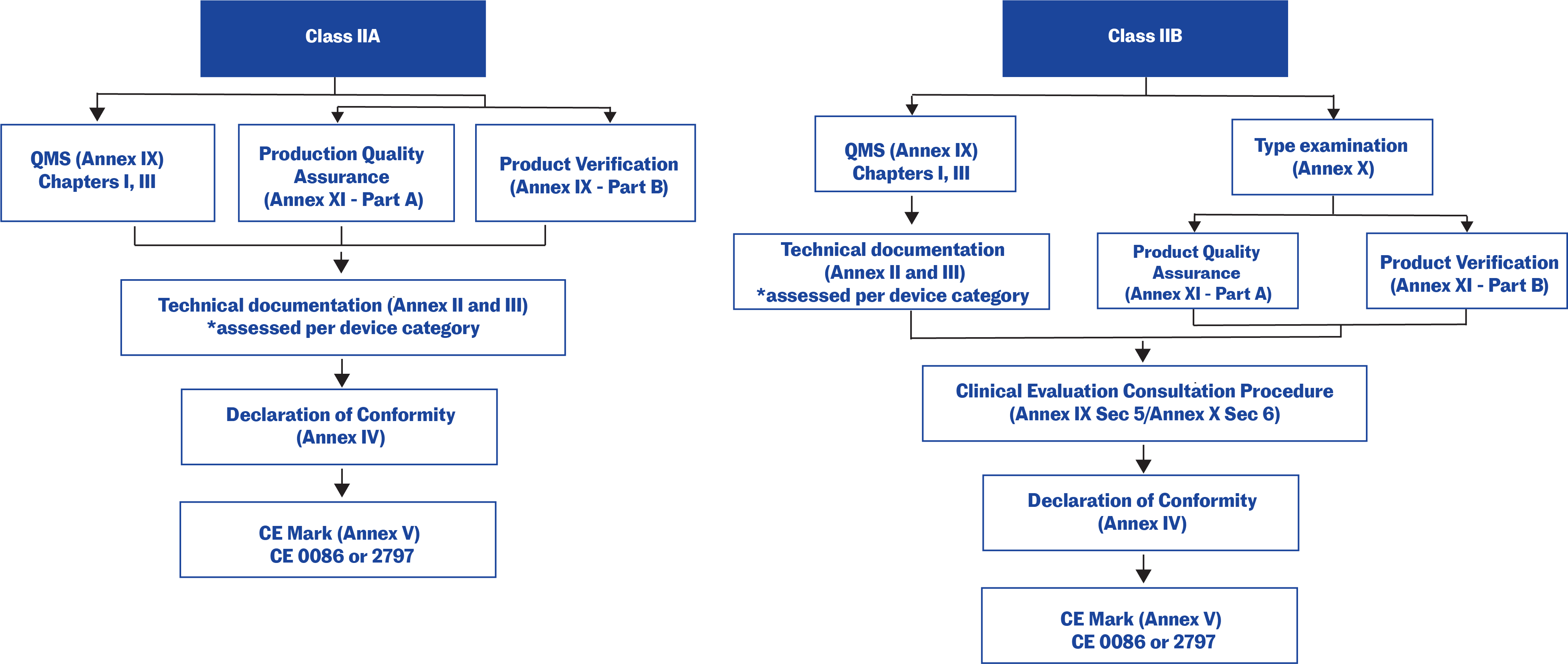
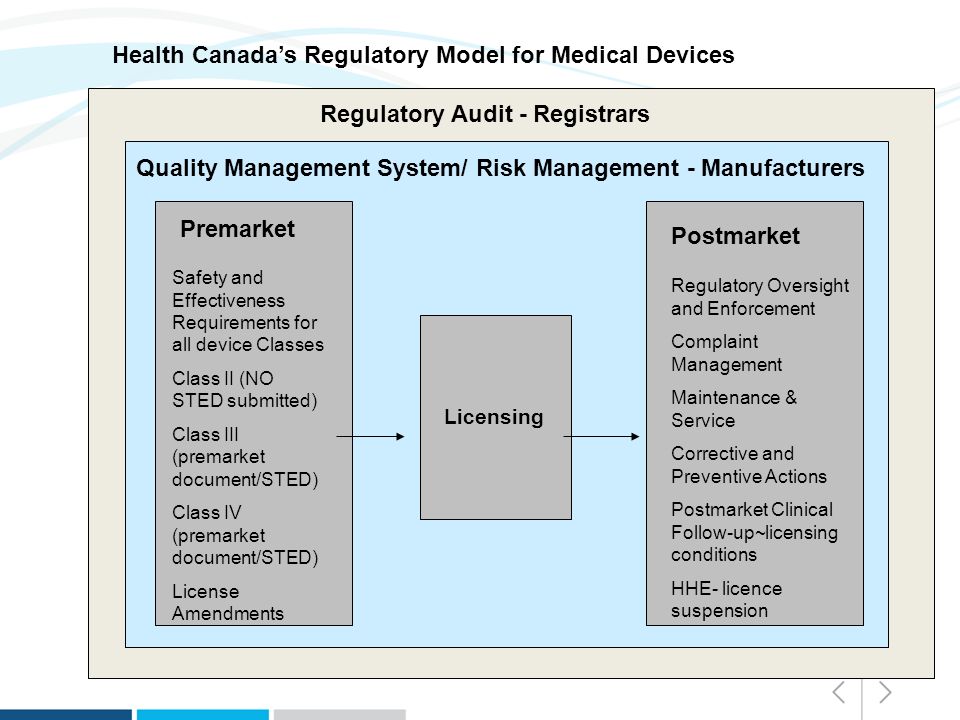
Overview of Medical Devices Regulations Velocity Science Sarah Chandler A/Manager, Device Licensing Services Division Medical Devices Bureau Health Canada. - ppt download

Case Study 28 : Registration of Class II Medical Device in Australia, Brazil, China, India, Pakistan and Russia

NSF International - Medical Devices e-Learning Portal | Canada Medical Device Regulations – A Comprehensive Overview (2 hours)

Overview of Health Canada regulations for medical devices and examples... | Download Scientific Diagram
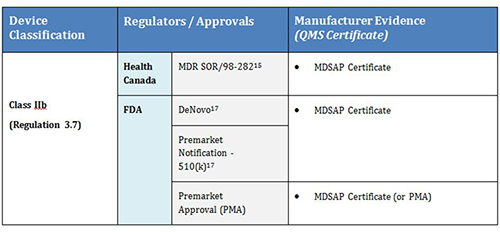
Use of market authorisation evidence from comparable overseas regulators / assessment bodies for medical devices (including IVDs) | Therapeutic Goods Administration (TGA)

Canadian Regulatory Amendments Introduce Additional Post-Market Surveillance Measures for Medical Devices
![PDF] A Policy Analysis to Inform Options for Medical Device Regulation and Post-Market Surveillance in the Canadian Context | Semantic Scholar PDF] A Policy Analysis to Inform Options for Medical Device Regulation and Post-Market Surveillance in the Canadian Context | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/9673644a2ed5a4fc07bb6532f1bb670f66c6780a/11-Table1-1.png)
PDF] A Policy Analysis to Inform Options for Medical Device Regulation and Post-Market Surveillance in the Canadian Context | Semantic Scholar

Canada 2018 Year in Review: Top 10 Medical Device Regulatory Issues - Food and Drug Law Institute (FDLI)

Canadian Good Manufacturing Practices: Pharmaceutical, Biotechnology, and Medical Device Regulations and Guidance Concise Reference (English Edition) eBook : Mindy J. Allport-Settle: Amazon.es: Tienda Kindle

Canada 2018 Year in Review: Top 10 Medical Device Regulatory Issues - Food and Drug Law Institute (FDLI)