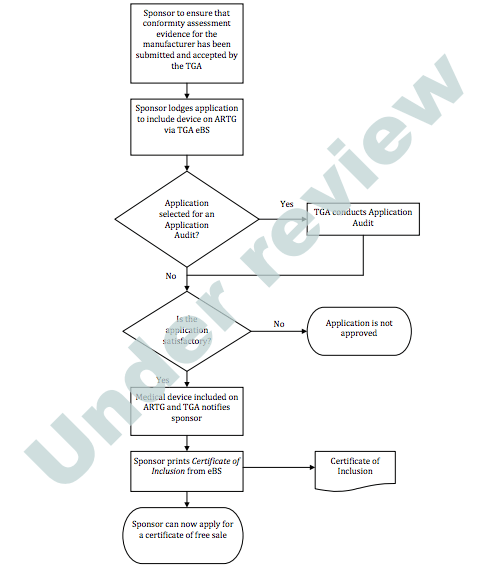
Prosthesis | Free Full-Text | Medical Device Regulation from a Health Service Provider's Perspective

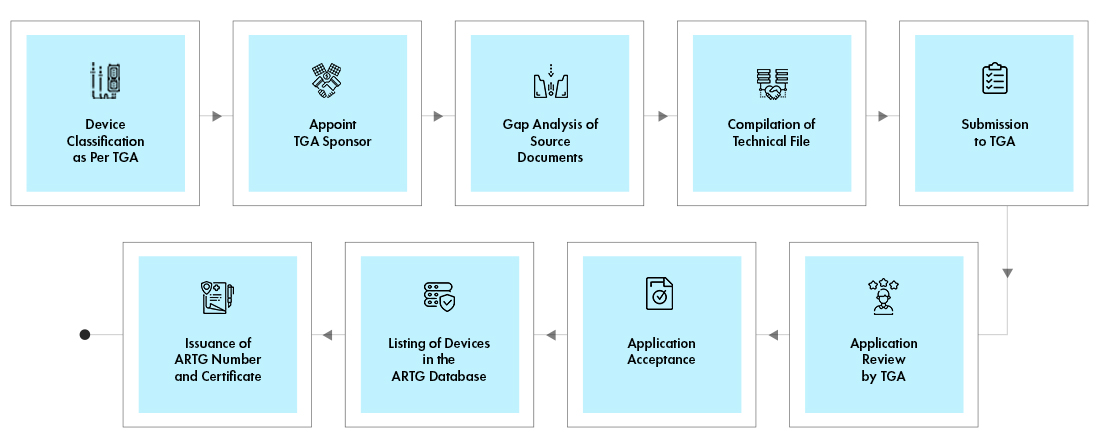
Newsletter January 2023 – Medical Device Regulations – update on Australian regulatory requirements for software based medical devices | Certification Body Australia (CBA)